
These findings suggest that this protein could be a key player in various fields, such as in detection and treatment of serious diseases (neurodegenerative diseases, cancer, and heart diseases, among others). Although BiP is still in early stages of study at a molecular level, some research groups have published findings of great value. BiP, a monomeric ATPase, has been referred to as the master regulator of the ER because of the broad and crucial roles that play in ER processes and functions, such as protein synthesis, folding, assembly, and translocation across the ER. One of the most important chaperones is BiP protein (immunoglobulin heavy-chain binding protein). Chaperones are proteins that assist other proteins in the folding process, facilitating correct folding pathways or providing microenvironments in which folding can occur. 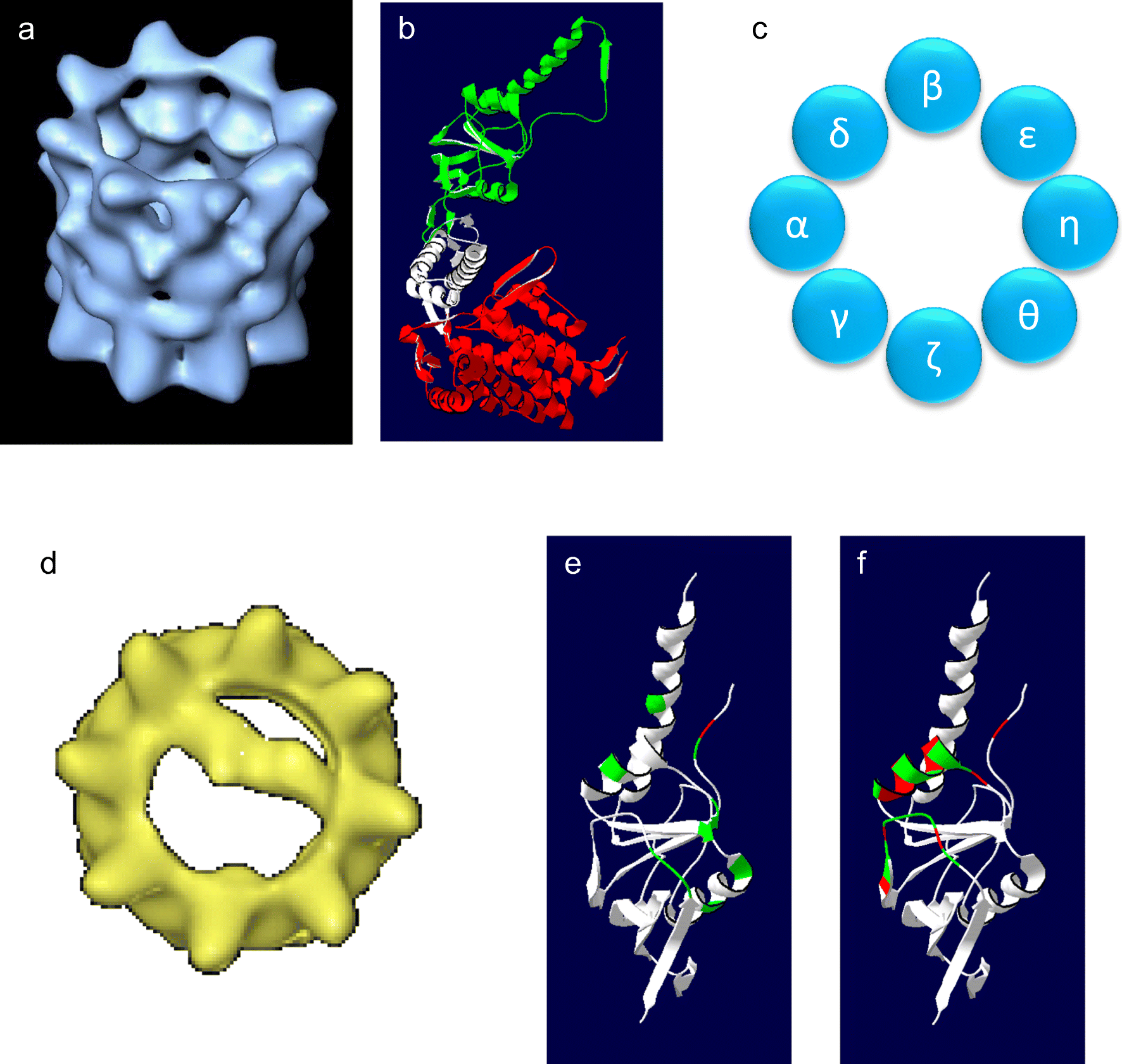
One of the most important functions of the ER involves the quality control (ERQC) of nascent proteins, which is accomplished by ER chaperones. The endoplasmic reticulum (ER) is involved in protein synthesis and the folding, assembly, transport, and secretion of nascent proteins. *Address all correspondence to: Introduction Biochemistry and Molecular Biology Department, Chemistry and Pharmaceutical Sciences Faculty, Mechanobiology and Single Molecule Biochemistry Laboratory, University of Chile, Chile.Structurally, BiP is formed by two domains, and to establish a correlation between BiP structure and catalysis and how its conformational and viscoelastic changes are coupled to ligand binding, catalysis, and allosterism (information transmitted between the domains), optical tweezers and nano-rheology techniques have been essential in this regard. One novel approach to study both structural and catalytic properties of BiP considers that the viscoelastic regime behavior of the enzymes (considering them as a spring) and their mechanical properties are correlated with catalysis and ligand binding. However, it is unknown what kind of molecular motor BiP works like, since the mechanochemical mechanism that BiP utilizes to perform its work during posttranslational translocation across the ER is not fully understood. In particular, it is involved in many endoplasmic reticulum (ER) processes and functions, such as protein synthesis, folding, and assembly, and also it works in the posttranslational mechanism of protein translocation. Its malfunction has been related with the appearance of many and important health problems such as neurodegenerative diseases, cancer, and heart diseases, among others. Immunoglobulin heavy-chain-binding protein (BiP protein) is a 75-kDa Hsp70 monomeric ATPase motor that plays broad and crucial roles maintaining proteostasis inside the cell.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
